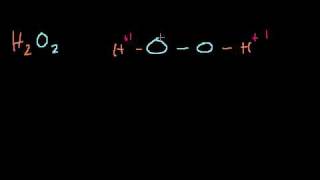
Spontaneous Combustion With H202 (Hydrogen Peroxide)
Thanks! Share it with your friends!
URL
Sorry, only registred users can create playlists.
Description
SUBSCRIBE!: http://bit.ly/1G97GmA
SHARE THIS VIDEO: https://youtu.be/HboqxpNUU94
Nickipedia Live Website: http://www.NickipediaLive.com
Team! I’ve always been told that 35% hydrogen peroxide is really dangerous. One of the dangers is that when H202 decomposes it creates oxygen gas (O2), which aids in making fires burn hotter, brighter, faster. This decomposition reaction also creates heat all by itself as an exothermic reaction! This is the perfect storm for spontaneous combustion!
We tried a cotton shirt and some dried grass. We were not disappointed with the results!
Follow Nickipedia
Twitter: http://www.twitter.com/nickuhas
Instagram: http://www.instagram.com/nickuhas
Facebook: http://www.facebook.com/uhasbrand
Nickipedia Website: http://www.NickipediaLive.com
Production Team:
Writer/Producer: Nick Uhas
Editor: Griffin Lewis
DP: Sam Mosco
SHARE THIS VIDEO: https://youtu.be/HboqxpNUU94
Nickipedia Live Website: http://www.NickipediaLive.com
Team! I’ve always been told that 35% hydrogen peroxide is really dangerous. One of the dangers is that when H202 decomposes it creates oxygen gas (O2), which aids in making fires burn hotter, brighter, faster. This decomposition reaction also creates heat all by itself as an exothermic reaction! This is the perfect storm for spontaneous combustion!
We tried a cotton shirt and some dried grass. We were not disappointed with the results!
Follow Nickipedia
Twitter: http://www.twitter.com/nickuhas
Instagram: http://www.instagram.com/nickuhas
Facebook: http://www.facebook.com/uhasbrand
Nickipedia Website: http://www.NickipediaLive.com
Production Team:
Writer/Producer: Nick Uhas
Editor: Griffin Lewis
DP: Sam Mosco
Post your comment
Comments
Be the first to comment